Medical connectors are different because medical applications are different. Medical systems require high reliability like industrial systems, but they have unique challenges where failure or unreliable operation can be life-threatening. A few examples include: Materials qualified for medical connectors are often specified to prevent bacteria growth and ensure that the connection system withstands sterilization or disinfection procedures. Connectors in medical systems may have to carry high voltages, optical links, fluids, and power and signal lines. Moisture resistance, cable management, and ergonomics are often important in medical equipment designs.
This FAQ begins with an overview of medical connector safety standards and related applications, presents hybrid cables that can include interconnects for signal, power, light, and fluids, covers the challenges associated with medical device misconnections, and closes with a review of recent developments related to small-bore connectors and luers.
IEC 60601-1 – “Medical electrical equipment – Part 1: General requirements for basic safety and essential performance” is an important document for specifiers of connectors for medical devices. The standard delineates two categories of protection:
- Means of Patient Protection (MOPP)
- Means of Operator Protection (MOOP)
IEC 60601-1 generally requires that two means of protection for both patients and operators be included in medical electronic devices; 2 MOPP and/or 2 MOOP. That ensures that if one means of protection fails, the second one still protects the user. Equipment or devices touching a patient when in use are required to have 2 MOPP. For equipment such as X-ray machines separated from the patient, 2 MOOP is considered sufficient to protect medical personnel.
Like all physical interconnects in electronic systems, connectors have creepage and clearance distances. The clearance distance is the shortest distance between two contacts through the air outside the solid insulation for connectors. The creepage distance is the shortest distance between two contacts along the surface of an insulation body. Connectors can achieve two means of protection by doubling the clearance and creepage distances between contacts (Table 1).

The level of protection required is directly related to the application. Three examples include:
- Type B (body) devices operate close to the body but without patient contact and must have 1,500 Vac insulation, 2.5mm creepage, 2.0mm clearance, and Basic insulation. X-ray machines, hospital beds, and MRI scanners are examples of type B devices.
- Type BF (body floating) devices are in physical contact with the patient and must have 3,000 Vac insulation, 5mm creepage, 4.0mm clearance, and Double insulation. Blood pressure monitors, thermometers, and ultrasound equipment are examples of type BF devices.
- Type CF (cardiac floating) devices are in physical contact with the heart and must have 4,000 Vac insulation, 8mm creepage, 5mm clearance, and Double insulation. Defibrillators and dialysis machines are examples of type CF devices.
Meeting IEC 60601-1 requires extended creepage and clearance distance that general-purpose commodity multipin connectors can’t match. Connectors meeting IEC60601-1 often have extended insulating collars or sleeves around the pins, or asymmetrical pin arrangements, to increase the creepage and clearance distances (Figure 1).

Hybrid medical connectors
As medical devices and equipment become increasingly complex and capable, cable management becomes a more important design aspect. The use of hybrid connectors provides one possible solution. Just as medical systems are getting more complex, so are hybrid medical connectors. Hybrid connectors can reduce device size and improve reliability and safety by eliminating the need for multiple cables and connectors. Hybrid connector systems eliminate the risk of cable entanglement, reduce cable stress, and speed connecting and disconnect medical devices. By eliminating the need for multiple connectors and cables, these interconnect systems enable clinicians or even patients to quickly and securely change umbilical or modular tools and handpieces that need connections to power, signal, and gases or liquids.
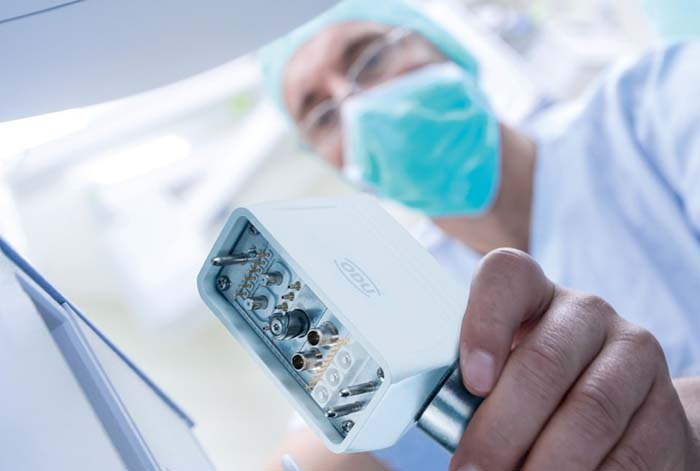
Users only have a single connector to check for proper mating instead of having to check multiple possible points of failure. And these connectors reduce the possibility of misconnections. In many environments such as intensive care units (ICUs), cardiac care units, emergency departments, even at home, patients may require several different devices at once, increasing the risk of misconnections.
Medical device misconnections
Medical device misconnections happen when two devices that perform different functions are connected in error or when a connection is not completely secured. While it’s possible to misconnect electrical connectors, misconnection is more common with connectors for liquids since there is a small variety of connectors for liquids compared with the breadth of electrical connector styles and formats. For long-term connections, fittings are often used. But for more common short-term uses, luers and quick disconnect couplings are the most frequently used tubing connectors in low-pressure environments. They are lightweight, cost-efficient, and, in the past, were mostly reliable. The ‘mostly reliable’ aspect could result in misconnections, possibly leading to patient injury or death. There are several possible reasons for misconnections:
- The limited number of sizes and shapes of low-pressure liquid connectors easily lead to confusion.
- Numerous sources of potential human error when connecting multiple devices to a patient including time pressure, inadequate lighting, unfamiliarity with a new system, and fatigue in high-stress environments.
Simple ‘fixes’ such as color-coding, tags, or labels are only somewhat successful. They can be helpful, but it’s still possible to physically misconnect devices. Due to the importance of ensuring secure and correct connections of low-pressure liquid fittings, industry standards have been developed.
ISO 594-2 – A partial solution to misconnections
The current ISO 80369 is a series of standards developed to improve on the shortcomings of ISO 594-2:1998 that was issued to address problems with misconnections in medical devices. ISO 594-2 references materials in the scope section with separate requirements for rigid and semi-rigid materials. Metal and glass are classified as rigid, while many plastics are semi-rigid. The standard does not address flexible and elastomeric materials. It does explicitly recognize that for fittings using semi-rigid materials (including most plastics), it was impossible to specify the fitting’s dimensions with sufficient accuracy. Fittings are still required to satisfy the functional/performance requirements.
It’s possible to design a male and female luer pair that meet the ISO 594-2 thread profile dimensions but which don’t work properly. Suppose a softer material such as nylon or polypropylene is used. Some users can over-rotate, or over-torque, the luer, causing damage to the threads resulting in an unsecured connection. The ISO 80369 series of standards were developed to address these concerns.
Evolving and Improving Small-Bore Connector Standards
Small-bore connectors have a connector opening diameter of less than 8.5 mm. They connect tubing, syringes, and other devices that carry fluids or gases for patient care. ISO 80369 is a series of standards to improve patient safety and reduce the risk of small-bore misconnections used in liquid and gas healthcare applications (Figure 3). For example, the new standard adds a specification for the “t” dimension for the male luer thread, defined as the distance from the end of the male luer to the first fully formed thread. Adding the “t” dimension helps to specify the thread profile dimensions more fully.

Two primary standards recognized by the FDA for small-bore connectors are:
ISO 80369-1:2010, “Small-bore connectors for liquids and gases in healthcare applications” – Part 1: General requirements – provides the methodology to assess non-interconnectable characteristics of small-bore connectors based on their inherent design and dimensions to reduce the risk of misconnections between medical devices
ISO 80369-20:2015, “Small-bore connectors for liquids and gases in healthcare applications” – Part 20: Common test methods – provides test methods to support small-bore connectors’ functional requirements.
ISO 80369 combines the semi-rigid and rigid dimensional requirements. It also adds a flexural modulus of 700 MPa or higher, not included in ISO 594. The addition of the flexural modulus makes it possible to define the dimensions with greater precision. For compliance, both the dimensional and functional requirements must be satisfied, even when using semi-rigid materials.
Where practical, ISO 80369 adopted some of the requirements in ISO 594. A luer that complies with ISO 80369 is backward compatible with ISO 594 requirements. However, a luer made from semi-rigid materials that passes only ISO 594 performance testing is incompatible with ISO 80369-7. ISO 80369 has expanded coverage to include several specific classes of devices:
80369-2 – Breathing systems and gas driving
80369-3 – Enteral applications such as feeding tubes
80369-5 – Limb cuff inflation devices such as blood pressure cuffs
80369-6 – Neuraxial applications such as epidural anesthesia catheters
80369-7 – Intravenous (IV) or hypodermic
In addition, a new section, “Part 2: Connectors for breathing systems and driving gases applications” is under development, and an additional part on “Connectors for urethral and urinary applications” is planned for the future.
Summary
Medical connectors are held to higher standards than industrial and other high-performance connectors. Proper design and use of medical connectors is needed to ensure the safety of patients and equipment operators. Hybrid connectors can minimize misconnections and enable users and patients to quickly and securely change umbilical or modular tools and handpieces that need connections to power, signal, and gases or liquids. The ISO 80369 is a series of standards developed to address problems with potentially dangerous misconnections when using medical devices, and ISO 80369 continues to evolve and expand its coverage.
References
ISO 80369 Small-Bore Connector Standards, Qosina
Medical Device Connectors, U.S. Food & Drug Administration
Reducing Risks through Standards Development for Medical Device Connectors, U.S. Food & Drug Administration
The IEC 60601-1 – Maximum patient and operator protection, ODU Connectors
The New ISO 80369-7 Luer Standard and How it Can Potentially Affect all Plastic Luer Users, Injectech



Leave a Reply
You must be logged in to post a comment.